
Lean Six Sigma Training Certification

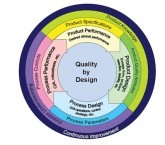
A long-standing axiom states that quality must not be “inspected in”, but rather “built in” if products and services are to have long-term success. Joseph Juran’s influential work, Juran On Quality By Design: The New Steps For Planning Quality Into Goods And Services (1992) exhaustively presents numerous cases studies from broad-ranging industries that show the vast majority of product and service failures are the result of poor design. At the turn of the millennium pharmaceutical products were defined largely by their manufacturing processes that were poorly understood, which produced products that were understood even less. Quality was determined almost entirely by post-process inspection, with virtually no foundation to build upon for corrective actions or continuous improvement. This state led to major global regulatory authorities for pharmaceuticals driving QbD initiatives over the last ten years that have become initiatives within the industry itself, where QbD practice has increasingly become Best Practice within many pharmaceutical companies.
The benefits of a Quality by Design approach to pharmaceutical products includes but is not limited to:
The practical application of QbD within the pharmaceutical space is a classic application of Design for Six Sigma principles and tools. The overall framework and tools used within QbD, e.g., risk-based quality management, statistical design of experiments (DoE), robust process design, and real-time process control through process analytical technology (PAT) as applied for pharmaceutical QbD efforts are areas of significant expertise for our instructors. The instructors will provide insight, instruction and application guidance for the QbD process and the tools required for its successful execution. Furthermore, they will also provide relevant insights and experiences they have gained through years of real-world QbD situations, as well as providing helpful context regard the current QbD landscape as it relates to current industry practices and regulatory agency interactions.
| Duration | Dates | Location | Cost | Course Code |
|---|
This material is available in an Adobe PDF that may be printed. To receive a copy, please send an email with “QBD” in the subject line to qbd@dmaic.us

Limited Time Discount
Save upto 20%
on ANY Lean Six Sigma Certification!
Turn workplace opportunities into impact.
Make the smart investment with our limited-time discounts on all Belt courses.
This will close in 0 seconds
"*" indicates required fields